- HOME
- CDMO Services
Globally acceptable“Knowledge”based on abundant experiences and
“Power”” generated by highly specialized technologies
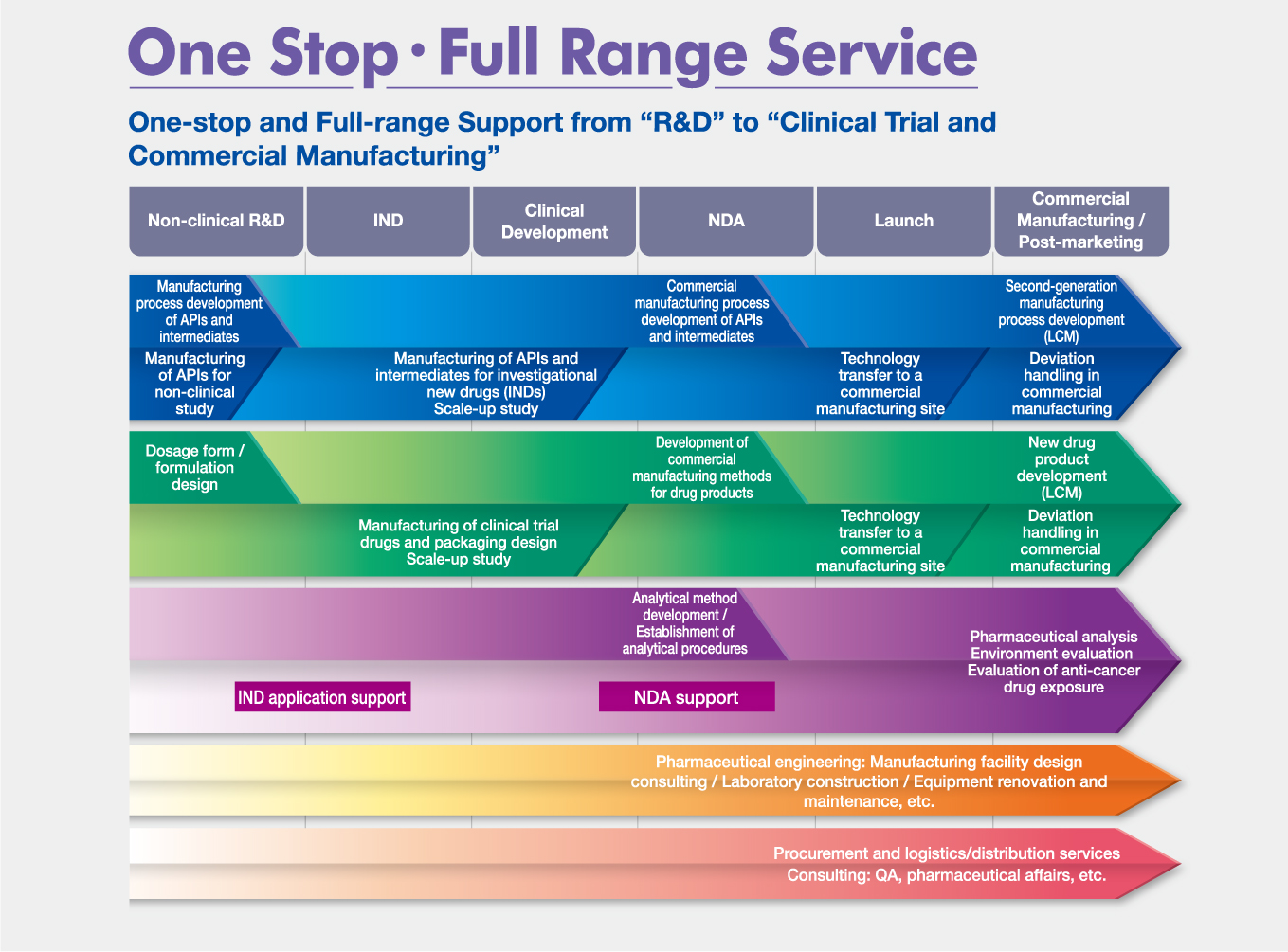
Shionogi Pharma can provide "Full-range Services" at "One Stop"
We can support our customers through our serivices including not only API process development, pharmaceutical development and commercial
manufacturing of API and drug products but also analytical method development, equipment design based on pharmaceutical engineering techlologies, etc.
Our sercvices will satisify our customers' needs.
Services
One-stop service from non-clinical stage to commercial
manufacturing and to LCM
One Stop Service
We can provide one-stop service covering every activity from non-clinical stage to commercial manufacturing and to Life Cycle Management.
High Quality
We manufacture quality drugs for clinical trials and commercial use at our five sites, which have successfully experienced inspections by the foreign and domestic health authorities.
Support
We support creation of various documents for applications and response to inquiries from the health authorities based on our abundant drug development experiences.
Engineering
We provide total engineering service covering basic design, validation and maintenance based on our experiences as a pharmaceutical manufacturer.
Full Range
We provide full-range service including development of analytical methods and their validation under three-region GMPs, environmental analyses and evaluation of anticancer drugs exposure at medical facilities.
Full Range Service from Shionogi Pharma
A tightly knit linkage between the various processes is very critical to ensure the success of new drug development and
to conduct manufacturing at a low cost.